Epigenetic control is critical during development, and frequently goes wrong in disease.
Imaging techniques such as super-resolution and lattice light sheet microscopy are enabling researchers to study precisely how epigenetic control is achieved. Understanding this process could lead to new ways of manipulating gene expression to treat disease.
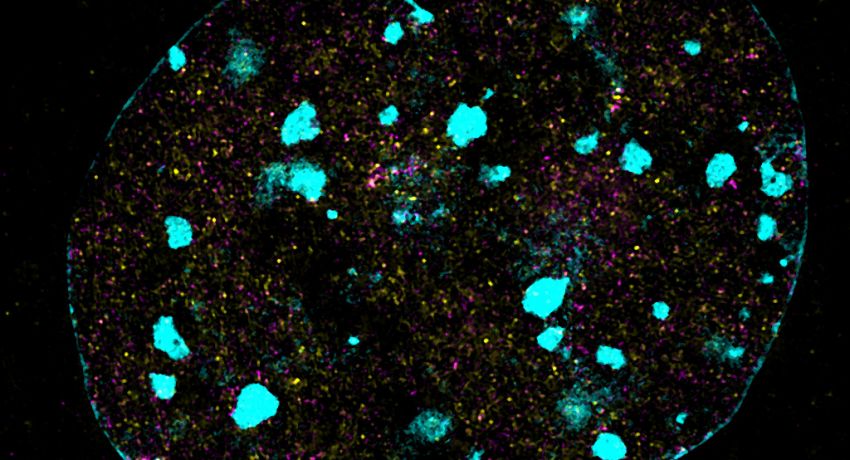
SIM (structural illumination microscopy) image of SMCHD1 in the nucleus, captured using Deltavision OMX SR. Credit: Iromi Wanigasuriya
Defects in SMCHD1 are associated with incurable diseases
Iromi Wanigasuriya, a PhD student in the lab of Associate Professor Marnie Blewitt, studies the protein SMCHD1 (Structural Maintenance of Chromosomes Flexible Hinge Domain Containing 1).
SMCHD1 is an epigenetic modifier that switches genes off when their expression is not required. SMCHD1 plays an important role in development, and defects in the protein are associated with several diseases including:
- Facioscapulohumeral muscular dystrophy (FSHD), a disease that causes progressive muscle wasting that first impacts patients in their teens or twenties;
- Bosma arhinia and micropthalimia syndrome (BAMS), a rare developmental disorder in which the nose fails to form; and
- Prader-Willi Syndrome, a complex disorder characterised by excessive appetite.
These diseases are debilitating and there are currently no targeted treatments available.
SMCHD1 function is poorly understood
Researchers understand relatively little about how SMCHD1 functions to switch genes off.
It is unclear how the protein interacts with chromatin in living cells, and how this interaction changes under different biological conditions.
New imaging techniques are revealing how SMCHD1 functions

The ability to observe live cell dynamics using imaging techniques such as airyscan, lattice light sheet and OMX SR have facilitated Ms Wanigasuriya to understand how SMCHD1 dynamics change in the cell in real time.
Ms Wanigasuriya is also exploring super-resolution techniques such as 3D SIM and dSTORM to investigate how SMCHD1 interacts with other proteins to form a large complex on DNA.
This information will help researchers understand how SMCHD1 is recruited to chromatin and how it functions to silence gene expression.
It is hoped these findings will lead to a better understanding of how various drugs affect SMCHD1’s structure and function in the nucleus.
Impact on new therapeutic approaches
A better understanding of how SMCHD1 functions will give researchers insights into how this goes awry in disease and potentially lead to new treatments for diseases involving SMCHD1 dysfunction.
Ms Wanigasuriya’s colleagues are currently screening for drugs that alter SMCHD1 activity to treat diseases such as FSHD. Advanced imaging techniques will be vital for studying how any potential drugs affect the dynamics of SMCHD1 in the nucleus and how it interacts with other proteins in a cellular context.